WHAT IS CIM-MEG19 ?

CIM-MEG19 is a scientifically validated formulation to enhance immunity to fight virus, bacteria, allergens and much more. CIM-MEG19 contains the standardised extract of Andrographis paniculata (Kalmegh) from selected varieties of plant and their parts. Andrographis has been shown to reduce duration and severity of upper respiratory infections such as those associated with the common cold or flu. It also reduced symptoms of rheumatoid arthritis. Andrographis paniculata is also used as astringent, bacteria killing agent, painkiller & fever reducer. Andrographis paniculata is also used for sore throat, coughs, swollen tonsils, bronchitis and allergies.
Who developed this medicine ?
CIM-MEG19 was developed by CSIR-CIMAP after years of research after seeing the global trend of Andrographis paniculata. CSIR-CIMAP is a leading research Institute of Council of Scientific and Industrial Research (CSIR), Government of India since 1959. It has a team of highly qualified scientists and a world class laboratory to accompany them in their research. Their core competency lies in developing standardised extract with respect to active phytochemicals in order to develop Phytopharmaceutical products. The traditional knowledge of Ayurvedic herbs is integrated with modern pharmacological evidences to develop quality products for global acceptance.

How are we sure that it works ?
CIM-MEG19 has been scientifically proven as a immunity booster which prevents cold and flu like conditions. A wide range of benefits and trials have been mentioned along with the scientific parameters adopted by CSIR-CIMAP which have been completed and found successful-
- In-vivo efficacy studies - Immunomodulatory (Immunity Booster)
- In-vivo efficacy studies - Antipyretic (Fever)
- In-vitro efficacy anti-inflammatory (redness, swelling, and pain) studies.
- In-vivo efficacy studies - Heptaoprotective pharmacological (Liver tonic)
- Animal toxicity and safety data (oral acute and sub-acute in mice)
- Genotoxicity- Ames test (-ve)
- Stability studies
- Force degradation studies
Its not just an Immunity Booster, it can also be taken for Ajirna (dyspepsia), Arsa (piles), Atisira (diarrhoea), Jvara (fever), Kanadu (itching), Kamala (jaundice), Kustha (diseases of skin); Prameha (increased frequency and turbidity of urine), Pravahika (dysentery), Tvakvikara (skin disorders); Vrana (wound), Yakrutvikara (disorders of liver).
Why only CIM-MEG19 ?
- No Synthetic Steroids or Hazardous Chemicals
- Natural herbs from captive cultivation
- Standardized Extract Derived From Specific Parts (Leaf, Stem & Fruits)
- Scientific validation done at CSIR-CIMAP– a renowned Research Institute of Government of India
- Pre-clinical Proven Efficacy of Hepatoprotection, Antipyretic, Aanalgesic, Anti-inflammatory and Immunity enhancing potential
- Defined Stability and Safety
- Each tablet ensured the presence of bioactives viz. Andrographolide, Neo-andrographolide, 14-Deoxy-11,12-didehydroandrographolide, and Andrograpnin
- Defined CMC (Chemistry, manufacturing and Controls)
- Standardization based on (04) marker for quality assurance
- Quality assurance from NABL accredited laboratory
- Continuous Monitoring for batch-to-batch variation
- Standardised quality formulation
Dosage:
For adult: Initially 1-2 tablet 1-3 times a day or as advised by the physician.
Use of KALMEGH (Andrographis paniculata) for respiratory disorders or Virus infecting Lungs:
- Ayurvedic description of pneumonia fever is defined as is called Raktasthivi Sanniat which severely affect respiratory system including and lungs.
- These symptoms are also being reported in the COVID-19 infected patients. Ayurvedic pharmacology defined such conditions primarily due to Pitta and Kaphaj-Vata which resulted as fever and profuse fluid in the lung(s).
- Available scientific research literature as well as recent WHO global research on coronavirus disease (COVID-19) also supports its utility as antiviral. Therefore, like modern medicines antiviral drugs, the repurposing of Andrographis seems to be beneficial in combating the severity of COVID-19 infection and /or related disorders. Though detail clinical studies are needed on this aspect. At present we do not have clinical data to claim. However, we have planned clinical efficacy studies to assess efficacy of CIM-MEG19 in COVID-19 patients in clinical improvement on WHO ordinal scale.
WHO: Global research on corona virus disease (COVID-19) pertaining to Kalmegh [2020-21]
- Andrographolide, a diterpenoid lactone compound of Andrographis paniculata, binds to lys353 and asp38 in the peptidase domain of human angiotensin-converting enzyme 2 [WHO COVID | ID: covidwho-1158820]
- Pharmacological actions of contents of Kabasura kudineer-a siddha formulation for fever with respiratory illness [WHO COVID | ID: covidwho-1140769]
- In-silico docking analysis revealed the potential of phytochemicals present in Phyllanthus amarus and Andrographis paniculata, used in Ayurveda medicine in inhibiting SARS-CoV-2 [WHO COVID | ID: covidwho-1023360]
- A pharmacology-based comprehensive review on medicinal plants and phytoactive constituents possibly effective in the management of COVID-19 [WHO COVID | ID: covidwho-911870]
- The coronavirus disease 2019 main protease inhibitor from Andrographis paniculata (Burm.f) Ness [WHO COVID | ID: covidwho-886254]
- Virtual screening of COVID-19 drug from three Indian traditional medicinal plants through in silico approach [WHO COVID | ID: covidwho-882033]
- The antiviral activity of andrographolide, the active metabolite from Andrographis paniculata (Burm. f.) wall. ex nees. against sars-cov-2 by using bio-and chemoinformatic tools [WHO COVID | ID: covidwho-830029]
- The Role of Adaptogens in Prophylaxis and Treatment of Viral Respiratory Infections [WHO COVID | ID: covidwho-829645]
- TGA advises of potential taste disturbance with Andrographis paniculata [WHO COVID | ID: covidwho-548168]
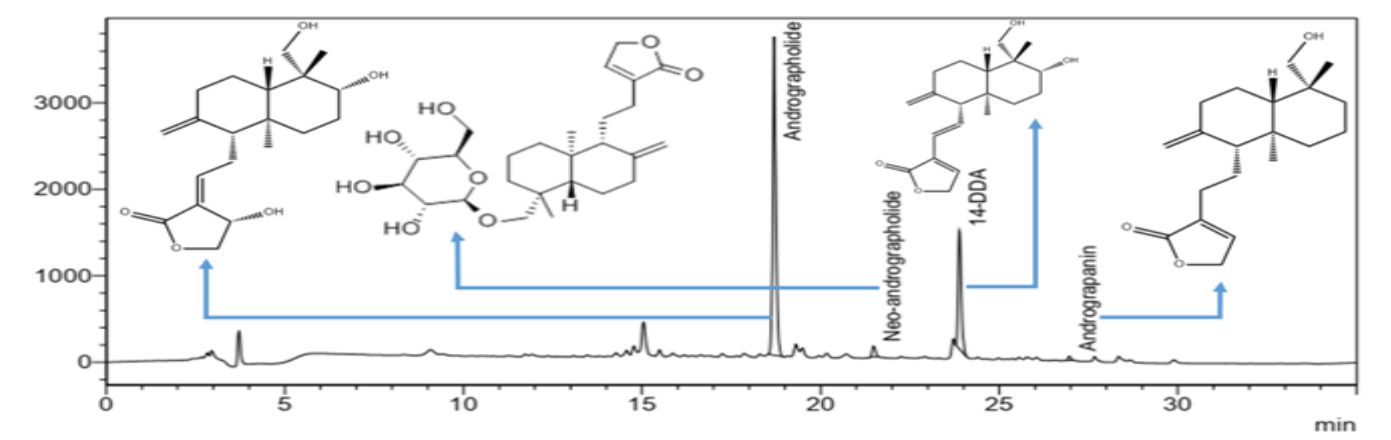 A representative chemical signature (HPLC chromatogram) of CIM-MEG19 tablet
A representative chemical signature (HPLC chromatogram) of CIM-MEG19 tablet
